Increased understanding of the function of the nanomaterial protein corona in driving nanomaterial uptake into, and impacts on, cells and organisms, and the resultant want for characterization of the corona, has led to a flourishing of strategies for isolation and evaluation of the constituent proteins over the previous decade.
However, regardless of over 700 corona research thus far, little or no is known in phrases of which strategies present probably the most exact and complete characterization of the corona. With the growing significance of the modeling of corona formation and its correlation with organic impacts, it’s well timed to correctly characterize and validate the isolation approaches used to find out the protein corona.
The present work introduces Capillary Electrophoresis with Electro Spray Ionization Mass Spectrometry (CESI-MS) as a novel technique for protein corona characterizations and develops an on-particle tryptic digestion technique, evaluating peptide solubilization options and characterizing the restoration of proteins from the nanomaterial floor.
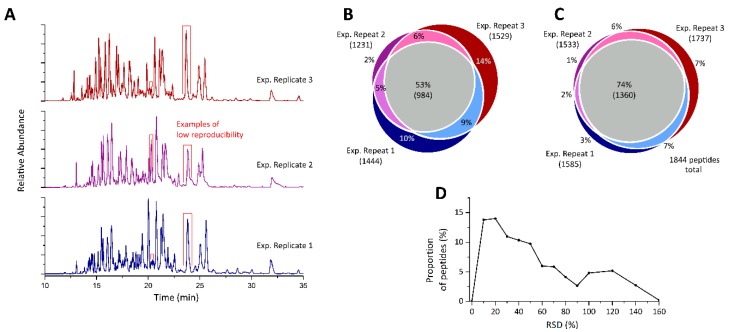
The CESI-MS was in comparison with the gold normal nano-LC-MS for corona evaluation and maintained a excessive diploma of reproducibility, whereas growing throughput by >3-fold. The on-particle digestion is in comparison with an in-solution digestion and an in-gel digestion of the protein corona. Interestingly, a variety of totally different protein courses had been discovered to be recovered to higher or lesser extents among the many totally different strategies.
Apolipoproteins had been detected at decrease concentrations when a surfactant was used to solubilize peptides, whereas immunoglobulins basically have a excessive affinity for nanomaterials, and thus present decrease restoration utilizing on-particle digestion.
The optimized on-particle digestion was validated utilizing 6 nanomaterials and proved succesful of recovering in extra of 97% of the protein corona.
These are necessary elements to contemplate when designing corona research and modeling corona formation and impacts, highlighting the importance of a complete validation of nanomaterial corona evaluation strategies.
Kinetics of Insertion and Folding of Outer Membrane Proteins by Gel Electrophoresis.
To look at the mechanisms of folding and insertion of TMPs into membranes, kinetic research are instrumental, for instance, for the evaluation of folding steps and concerned intermediates or for the dedication of activation energies. For many β-barrel transmembrane proteins (β-TMPs) it has been proven that the folded, purposeful kind will be separated from the unfolded kind by a easy electrophoretic mobility assay.
The solely necessities for a separation by SDS-polyacrylamide gel electrophoresis (SDS-PAGE) are that the folded kind is sufficiently secure and that the samples should not heat-denatured earlier than the electrophoresis is carried out. Many folded β-TMPs resist the therapy with SDS at room temperature and are secure in opposition to forces throughout electrophoresis.
On the opposite facet, SDS additionally binds to unfolded varieties of β-TMPs and prevents their folding into β-barrel construction. These observations have been used to develop a easy assay to watch the kinetics of β-barrel tertiary construction formation in a membrane surroundings by electrophoresis.
A folding response of a β-TMP is initiated by dilution of the denaturant within the presence of preformed lipid bilayers, proteoliposomes or membrane vesicles. At chosen instances, samples are taken from the response. In these samples, folding is stopped by addition of SDS.
At the tip of the complete folding response, all samples are analyzed by SDS-PAGE and the fractions of folded β-TMP that they comprise are decided by densitometry.
An benefit of this kinetic assay is that it not solely permits a direct dedication of fractions of folded and unfolded varieties at a specific time throughout folding of the β-TMP right into a membrane, but in addition facilitates the dedication of the influence of folding elements (e.g., molecular chaperones) or folding equipment that the majority typically have a special molecular mass and electrophoretic mobility.
The assay has been very helpful to look at how folding and insertion is affected by the construction of the phospholipids within the lipid bilayer and the way folding equipment compensates for the presence of membrane lipids that retard folding and insertion of β-TMPs.